Lithium-ion Battery Materials & Manufacturing
A comprehensive guide to the components, materials, and production processes that power modern energy storage solutions
Lithium-ion batteries have revolutionized portable electronics, electric vehicles, and renewable energy storage. To understand what is in a lithium ion battery, we must examine both the raw materials that compose these advanced energy storage devices and the intricate manufacturing processes that transform these materials into functional batteries. What is in a lithium ion battery goes beyond just lithium - it's a sophisticated combination of various materials working in harmony to store and release energy efficiently.
This detailed exploration will delve into the core components that answer the question, "what is in a lithium ion battery," including cathode materials, anode materials, electrolytes, separators, and various auxiliary components. Additionally, we'll examine the complete manufacturing process that turns these raw materials into the high-performance batteries powering our modern world.
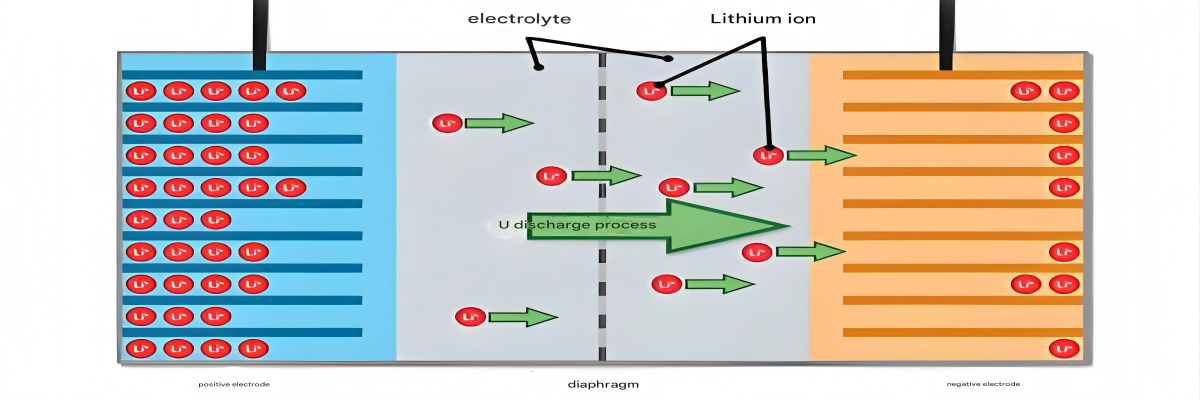
The Composition of Lithium-ion Batteries
To truly understand what is in a lithium ion battery, we need to break down its components into their fundamental materials. Each part plays a critical role in the battery's performance, safety, and longevity, from the active materials that store energy to the separators that prevent short circuits.
Key Materials in Lithium-ion Batteries
Positive Electrode Compositions
When exploring what is in a lithium ion battery, the cathode materials are among the most critical components, as they largely determine the battery's energy density and voltage. These are typically micron-scale powder materials that enable the electrochemical reactions necessary for energy storage.
Commercially available cathode materials include several key formulations, each with distinct properties suited for specific applications. What is in a lithium ion battery's cathode can vary significantly based on intended use, from consumer electronics to electric vehicles.
- Lithium Cobalt Oxide (LiCoO₂): Widely used in 3C battery applications (computers, communications, and consumer electronics) due to its high energy density.
- Lithium Manganese Oxide (LiMn₂O₄): Known for its thermal stability and lower cost compared to cobalt-based alternatives.
- Ternary Materials (LiNiₓMnᵧCo_zO₂): Combine nickel, manganese, and cobalt in varying proportions to balance energy density, stability, and cost.
- Lithium Iron Phosphate (LiFePO₄): Valued for its safety, long cycle life, and lower environmental impact, commonly used in electric vehicles and energy storage systems.

Negative Electrode Compositions
The anode is another essential component when considering what is in a lithium ion battery, responsible for storing lithium ions during charging. Modern batteries utilize various anode materials, each with distinct advantages in terms of capacity, conductivity, and cycling stability.
What is in a lithium ion battery's anode has evolved significantly, with graphite remaining the most common material due to its reliability and performance characteristics. However, research continues to develop next-generation anode materials to improve battery performance.
- Graphite Materials: The most widely used anode materials, offering good conductivity and stable cycling performance.
- Hard Carbon Materials: Amorphous carbon structures with higher capacity potential than traditional graphite.
- Soft Carbon Materials: Graphitizable carbons with good electrical conductivity.
- Lithium Titanate: Offers excellent safety and long cycle life, though with lower energy density.
- Silicon-based (Si) Materials: Promising high-capacity alternatives under development.
- Tin-based (Sn) Materials: Another high-capacity option being researched for future battery technologies.
Ion Conducting Mediums
Understanding what is in a lithium ion battery must include the electrolyte, the medium that facilitates the movement of lithium ions between the cathode and anode during charging and discharging. The electrolyte's composition significantly impacts battery performance, safety, and operating temperature range.
What is in a lithium ion battery's electrolyte typically includes lithium salts dissolved in organic solvents, creating a conductive medium for ion transport. Recent developments have also introduced gel electrolytes as alternatives to traditional liquid formulations.
Common Electrolyte Components:
Lithium Salts:
Lithium hexafluorophosphate (LiPF₆) is the most commonly used salt, providing good ionic conductivity and stability.
Organic Solvents:
Mixtures typically include ethylene carbonate (EC), dimethyl carbonate (DMC), diethyl carbonate (DEC), and ethyl methyl carbonate (EMC).
The specific combination of solvents is carefully formulated to balance viscosity, conductivity, and electrochemical stability, ensuring optimal performance across different operating conditions.
Ion-Permeable Barriers
When examining what is in a lithium ion battery, the separator plays a crucial but often overlooked role as a physical barrier between the cathode and anode. This component prevents direct electrical contact (which would cause a short circuit) while allowing the passage of lithium ions.
What is in a lithium ion battery's separator is typically a porous polymeric material designed to provide high ion permeability, mechanical strength, and thermal stability. The separator's structure and material composition are critical factors in battery safety and performance.
| Separator Type | Material | Characteristics |
|---|---|---|
| Single-layer porous membrane | Polyethylene (PE) | Good mechanical properties and thermal shutdown capability |
| Single-layer porous membrane | Polypropylene (PP) | Higher melting point than PE, good chemical resistance |
| Multi-layer porous membrane | PP/PE/PP | Combines advantages of both materials, enhanced safety |

Protective Enclosures
The outer casing is another important element when considering what is in a lithium ion battery, providing physical protection, structural integrity, and in some cases, thermal management. The choice of casing material depends on the battery's intended application, size, and performance requirements.
What is in a lithium ion battery's casing can vary significantly between different form factors and applications, from flexible pouches for consumer electronics to rigid metal enclosures for electric vehicles.
Aluminum-Plastic Composite Films
Used primarily in pouch-type batteries, offering flexibility, light weight, and good barrier properties against moisture and gases.
Aluminum Casings
Common in prismatic and cylindrical batteries, providing good structural strength while remaining relatively lightweight.
Stainless Steel Casings
Offering superior strength and corrosion resistance, used in applications requiring enhanced durability and safety.

Supporting Components
To fully answer what is in a lithium ion battery, we must consider the various auxiliary materials that enable proper functioning and performance. These components, while not directly involved in energy storage, play vital roles in conductivity, structural integrity, and overall battery performance.
Conductive Agents
These materials enhance electron conductivity within the electrode structures, ensuring efficient current collection. What is in a lithium ion battery's conductive agent formulation can include:
- Carbon black (various types including acetylene black)
- Vapor-grown carbon fibers (VGCF)
- Carbon nanotubes
- Graphene and graphene derivatives
Binders
Binders hold the active materials, conductive agents, and current collectors together, maintaining electrode structure during cycling. Common binders include:
- Polyvinylidene fluoride (PVDF) - used for both cathodes and anodes
- Styrene-butadiene rubber (SBR) - typically used for anodes
- Carboxymethyl cellulose (CMC) - often used with SBR in anode formulations
Current Collectors & Tabs
These components collect and conduct electrons from the electrodes to the external circuit:
- Cathode current collector: Aluminum foil
- Cathode tabs: Aluminum sheets
- Anode current collector: Copper foil
- Anode tabs: Nickel sheets

Lithium-ion Battery Manufacturing Process
A detailed look at the sophisticated production processes that transform raw materials into high-performance batteries
Electrode Preparation
The manufacturing process begins with the preparation of electrode slurries. For both positive and negative electrodes, active powder materials are mixed with binders, solvents, and conductive agents.
This mixture undergoes thorough stirring and dispersion to ensure uniform distribution of all components. The resulting slurry is then evenly coated onto current collectors (aluminum foil for cathodes, copper foil for anodes) and dried to remove solvents.
After drying, the electrodes undergo calendering (roller pressing) to achieve the desired thickness and density, followed by slitting to produce electrodes of the required dimensions.
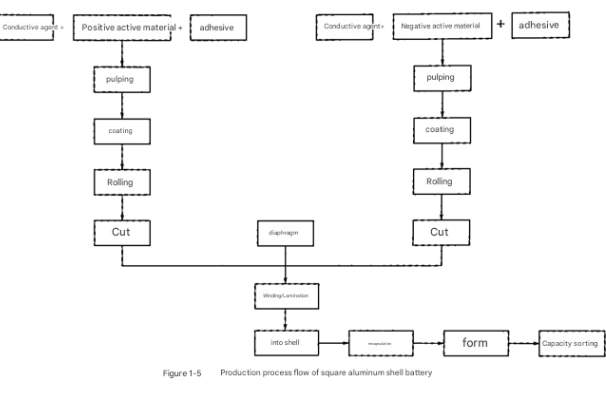
Cell Assembly
The assembly process starts with welding positive and negative tabs to their respective electrodes. These tabs facilitate electrical connection to the external terminals.
Electrodes are then combined with separators in either a winding or stacking process to form the cell core. The winding process rolls the electrodes and separators into a cylindrical or prismatic shape, while stacking layers them alternately for pouch or prismatic designs.
The assembled cell core is then enclosed in its housing - either aluminum-plastic composite film for pouch cells, aluminum casings for prismatic cells, or stainless steel casings for certain cylindrical designs.
Electrolyte Filling
Before electrolyte filling, the assembled cells undergo a drying process to remove any moisture, which can react with the electrolyte and degrade battery performance.
The electrolyte is then injected into the cell in a controlled, moisture-free environment (glove box or dry room). This step requires precision to ensure proper infiltration of the electrolyte into all porous structures within the cell.
After filling, the cell is partially sealed to allow for gas release during subsequent formation processes.


Formation and Aging
Formation is a critical process where the battery undergoes its first charge, creating the solid electrolyte interphase (SEI) layer on the anode. This layer is essential for long-term battery performance and cycle life.
The formation process is carefully controlled with specific charging rates and voltage limits to ensure proper SEI formation. Gases generated during this process are vented before the cell is fully sealed.
Following formation, cells undergo an aging process, where they are stored at controlled temperatures for a specified period. This allows for stabilization of electrochemical properties and identification of early failures.
Grading and Sorting
After aging, batteries undergo comprehensive testing to evaluate their performance characteristics. This process, known as grading or sorting, ensures consistent quality and performance.
Key parameters measured include capacity, internal resistance, voltage characteristics, thickness, and dimensional accuracy. These tests help answer what is in a lithium ion battery in terms of its performance capabilities.
Batteries are then categorized into different grades based on these parameters. This sorting is crucial for applications requiring multiple cells, as it ensures balanced performance in battery packs.


Final Packaging
After grading, batteries that meet quality standards proceed to final packaging. This involves placing cells in protective enclosures, attaching connectors where necessary, and applying labels with important information.
For applications requiring multiple cells, this stage may include assembling cells into modules or complete battery packs, along with necessary electronics for monitoring and balancing.
Final quality checks ensure that all products meet specifications before they are prepared for shipment. This completes the transformation from raw materials to finished lithium-ion batteries ready for integration into various devices and systems.
Summary of Lithium-ion Battery Composition
Understanding what is in a lithium ion battery reveals a sophisticated combination of materials working together to provide efficient energy storage. From active electrode materials to supporting components, each element plays a crucial role in determining the battery's performance characteristics.
What is in a lithium ion battery extends beyond its active materials to include carefully engineered electrolytes, separators, and casing materials, all manufactured through precise processes to ensure reliability, safety, and performance. As technology advances, researchers continue to innovate both the materials and manufacturing techniques to create batteries with higher energy density, longer cycle life, and improved safety profiles.Electronic shelf labels.
The manufacturing process transforms these raw materials through several critical stages, each requiring careful control and precision to produce high-quality batteries. From electrode preparation to final testing, each step contributes to the final performance of the battery.Related Hydraulic Spare Parts.