Lithium Ion Battery Principles
A comprehensive guide to the electrochemical mechanisms and structural design of the modern lithium battery
The lithium battery has revolutionized portable electronics, electric vehicles, and renewable energy storage since its commercial introduction in 1991 by Sony. Unlike traditional batteries, the lithium battery operates on the principle of ion migration rather than electron transfer in chemical reactions, offering higher energy density, longer cycle life, and lighter weight. This page explores the fundamental electrochemical principles and structural components that make the lithium battery the preferred energy storage solution in today's technology-driven world.

Understanding the lithium battery requires knowledge of both its electrochemical processes and physical construction. The intricate balance between material science and electrochemistry enables the lithium battery to deliver consistent power while maintaining stability through hundreds, even thousands, of charge-discharge cycles. This guide examines these components in detail, from atomic-level reactions to macroscopic cell designs.
Fundamental Lithium Battery Reactions
The operation of a how do lithium ion batteries work is based on the reversible intercalation and deintercalation of lithium ions between two electrodes during charge and discharge cycles. This process distinguishes the lithium battery from other battery technologies that rely on chemical reactions involving mass transport of active materials. In a lithium battery, the lithium ions (Li⁺) act as charge carriers, moving between the positive and negative electrodes through an electrolyte while electrons flow through an external circuit to provide power.
Key Electrochemical Reactions
During discharge, the lithium battery undergoes the following half-reactions:
- Anode (Oxidation): LiC₆ → Li⁺ + C₆ + e⁻
- Cathode (Reduction): Li₁₋ₓCoO₂ + xLi⁺ + xe⁻ → LiCoO₂
- Overall Reaction: LiC₆ + Li₁₋ₓCoO₂ → C₆ + LiCoO₂
During charging, these reactions reverse, with lithium ions migrating back to the anode where they intercalate into the carbon structure.
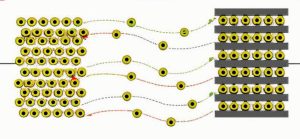
Lithium ion migration in a lithium battery during charge-discharge cycles
Intercalation Mechanism
A critical feature of the lithium battery is the intercalation process, where lithium ions insert themselves into the crystal structure of the electrode materials without significantly altering the host structure. This intercalation allows for reversible charging and discharging, which is essential for the long cycle life of a lithium battery.
The host materials must maintain structural integrity during ion insertion and extraction. This stability is what enables the lithium battery to undergo hundreds of cycles while retaining its capacity, unlike batteries that rely on phase changes or dissolution-precipitation reactions.
Electrochemical Potential
The voltage of a lithium battery is determined by the difference in electrochemical potential between the anode and cathode materials. Lithium has a very low reduction potential (-3.04 V vs. standard hydrogen electrode), which contributes to the high voltage of the lithium battery compared to other chemistries. Most commercial lithium battery cells produce between 3.0 and 3.7 volts, significantly higher than the 1.5 volts of alkaline batteries.
Charge-Discharge Kinetics
The performance of a lithium battery depends heavily on the kinetics of lithium ion transport and electron transfer at the electrode-electrolyte interfaces. Several factors influence these kinetics:
- Ionic conductivity of the electrolyte, which determines how quickly lithium ions can move between electrodes
- Diffusion coefficient of lithium ions within the electrode materials, affecting how rapidly ions can intercalate
- Electrode surface area, which influences the rate of charge transfer reactions
- Charge transfer resistance at the electrode-electrolyte interface
These kinetic factors determine the lithium battery's rate capability—how quickly it can be charged or discharged while maintaining stable operation and capacity.
Thermodynamics of Lithium Battery Systems
The energy storage capacity of a lithium battery is fundamentally a thermodynamic property determined by the Gibbs free energy change (ΔG) of the cell reaction. The relationship between the Gibbs free energy and the cell voltage (E) is given by the equation:
ΔG = -nFE
where n is the number of moles of electrons transferred, and F is Faraday's constant (96,485 C/mol)
For a lithium battery, this relationship means that materials with a larger difference in their electrochemical potentials will produce a higher voltage and thus greater energy density. This thermodynamic principle drives the selection of electrode materials in lithium battery design.
Energy and Power Density
The lithium battery achieves high energy density through a combination of high voltage and high capacity. Energy density (amount of energy stored per unit mass) is calculated as the product of voltage and capacity. Power density (rate of energy delivery) depends on how quickly the electrochemical reactions can proceed.
These two parameters—energy density and power density—represent a fundamental trade-off in lithium battery design. Applications like electric vehicles require both high energy density (for range) and high power density (for acceleration), challenging material scientists to develop optimized electrode formulations.

Energy density comparison of various battery technologies, with lithium battery leading
Entropy Changes in Lithium Battery Operation
The temperature dependence of a lithium battery's voltage arises from entropy changes (ΔS) during the cell reaction, described by the Gibbs-Helmholtz equation. This temperature effect is important for lithium battery management systems, as operating temperatures significantly impact performance and safety.
Exothermic reactions during discharge can increase cell temperature, potentially leading to thermal runaway if not properly managed. This is a critical consideration in lithium battery design and has led to the development of sophisticated thermal management systems in applications ranging from smartphones to electric vehicles.
Degradation Mechanisms in Lithium Battery
Over time, all lithium battery cells experience capacity fade and performance degradation due to various electrochemical and chemical processes. Understanding these mechanisms is crucial for improving lithium battery longevity and reliability.
SEI Layer Formation
The Solid Electrolyte Interface (SEI) forms on the anode surface during the first charge cycle of a lithium battery. While a stable SEI is essential for preventing further electrolyte decomposition, excessive growth consumes lithium ions and reduces capacity over time.
Electrode Degradation
Repeated expansion and contraction of electrode materials during charge-discharge cycles can cause structural damage in a lithium battery. This mechanical stress leads to particle cracking, loss of electrical contact, and reduced ion diffusion pathways.
Lithium Plating
At high charge rates or low temperatures, lithium ions may deposit as metallic lithium on the anode surface instead of intercalating into the structure. This lithium plating reduces capacity and creates safety hazards in a lithium battery.
Electrolyte Degradation
Electrolyte decomposition reactions, accelerated by high temperatures, produce gases and consume active materials in a lithium battery. This reduces ionic conductivity and can lead to increased internal resistance and cell swelling.

Microscopic view of electrode degradation in a cycled lithium battery
Cycle Life and Calendar Aging
The cycle life of a lithium battery refers to the number of charge-discharge cycles it can undergo before its capacity drops to 80% of its initial value. This typically ranges from 500 to 2000 cycles for consumer lithium battery products, though advanced designs can exceed 5000 cycles.
Calendar aging—capacity loss over time even when not in use—affects all lithium battery cells. This process is accelerated by high temperatures and high state-of-charge storage conditions. Proper storage practices (cool temperatures, ~50% charge) can significantly extend a lithium battery's shelf life.
Basic Lithium Battery Cell Structure
A lithium battery cell consists of four primary components working together to enable the electrochemical reactions: two electrodes (anode and cathode), a separator, and an electrolyte. These components are carefully engineered to maximize the lithium battery's performance, safety, and longevity.

Exploded view of a lithium battery cell showing key components
Component Functions
Anode
The anode (negative electrode) of a lithium battery serves as the host material for lithium ions during charge. Most commercial lithium battery anodes are made of graphite, a form of carbon with a layered structure that efficiently accommodates lithium ions through intercalation. Advanced lithium battery designs are exploring silicon-based anodes, which offer much higher capacity but present challenges with volume expansion.
Cathode
The cathode (positive electrode) provides the source of lithium ions in a lithium battery. Cathode materials determine many key characteristics of the lithium battery, including voltage, energy density, and safety. Common cathode materials include lithium cobalt oxide (LCO), lithium nickel manganese cobalt oxide (NMC), lithium iron phosphate (LFP), and lithium manganese oxide (LMO).
Electrolyte
The electrolyte in a lithium battery facilitates the movement of lithium ions between the anode and cathode while preventing electron flow that would short-circuit the cell. Traditional lithium battery electrolytes are liquid solutions containing lithium salts (typically LiPF₆) dissolved in organic solvents like ethylene carbonate and dimethyl carbonate. Solid-state lithium battery technologies use solid electrolytes, offering improved safety and potentially higher energy density.
Separator
The separator is a porous membrane that physically separates the anode and cathode in a lithium battery to prevent electrical short circuits while allowing lithium ions to pass through. Most separators are made from polyolefin materials like polyethylene or polypropylene. The separator's porosity, thickness, and mechanical strength are critical factors in lithium battery performance and safety, particularly under thermal or mechanical stress.
Current Collectors
Current collectors are conductive materials that collect electrons from the anode and deliver them to the cathode in a lithium battery. The anode current collector is typically a copper foil, while the cathode uses aluminum foil, chosen for their conductivity, corrosion resistance, and cost-effectiveness. These thin foils minimize weight while maximizing electron flow, contributing to the overall efficiency of the lithium battery.
Lithium Battery Cell Form Factors
Lithium battery cells are manufactured in various form factors to accommodate different applications, from small portable electronics to large energy storage systems. Each design offers distinct advantages in terms of energy density, power delivery, mechanical flexibility, and manufacturing cost.

Cylindrical Cells
Cylindrical lithium battery cells feature a metal casing (typically steel) in standardized sizes like 18650 (18mm diameter, 65mm length) and 21700. Their robust construction offers good thermal performance, making them popular in laptops, power tools, and electric vehicles.

Prismatic Cells
Prismatic lithium battery cells use a rectangular aluminum or plastic casing, offering efficient space utilization. This design is common in smartphones, tablets, and electric vehicles where packaging efficiency is critical.

Pouch Cells
Pouch lithium battery cells use a flexible, lightweight polymer casing, providing the highest energy density by weight. Their flexibility allows for custom shapes, making them ideal for thin devices and wearable technology.
Module and Pack Assembly
For applications requiring higher voltage or capacity than a single lithium battery cell can provide, multiple cells are combined into modules, which are then assembled into packs. This hierarchical structure allows for flexible scaling of lithium battery systems.
Lithium battery packs include additional components beyond the cells themselves:
- Battery Management System (BMS): Monitors and controls cell balancing, temperature, voltage, and current
- Thermal management system: Maintains optimal operating temperatures
- Mechanical structure: Provides physical protection and mounting points
- Electrical connections: Distribute current and enable communication
These components ensure safe, efficient operation of the entire lithium battery system, particularly critical in large applications like electric vehicles and grid storage.

Electric vehicle lithium battery pack with cells, cooling system, and BMS
Lithium Battery Chemistries and Classifications
Lithium battery technology encompasses several distinct chemistries, primarily differentiated by their cathode materials. Each chemistry offers unique characteristics that make it suitable for specific applications, balancing factors like energy density, power output, safety, cost, and cycle life.
| Chemistry | Abbreviation | Key Characteristics | Typical Applications |
|---|---|---|---|
| Lithium Cobalt Oxide | LCO | High energy density, moderate cycle life, lower thermal stability | Smartphones, laptops, portable electronics |
| Lithium Nickel Manganese Cobalt Oxide | NMC | Balanced energy/power, good cycle life, improved safety | Electric vehicles, power tools, energy storage |
| Lithium Iron Phosphate | LFP | Excellent safety, long cycle life, lower energy density | Electric vehicles, stationary storage, marine applications |
| Lithium Manganese Oxide | LMO | High power, good thermal stability, lower cycle life | Hybrid vehicles, medical devices, power tools |
| Lithium Nickel Cobalt Aluminum Oxide | NCA | Very high energy density, good power, moderate safety | Electric vehicles, drones, high-performance applications |
Emerging Lithium Battery Technologies
Research and development continue to advance lithium battery technology, with several promising approaches on the horizon:
Solid-State Batteries
Replacing liquid electrolytes with solid materials could revolutionize the lithium battery by offering higher energy density, faster charging, and significantly improved safety. Solid-state lithium battery technologies eliminate flammable liquid electrolytes and enable the use of lithium metal anodes.
Silicon Anode Batteries
Silicon can store up to 10 times more lithium than graphite, potentially dramatically increasing lithium battery energy density. Challenges with volume expansion during cycling are being addressed through nanostructuring and composite material approaches.
Lithium-Sulfur Batteries
Lithium-sulfur chemistry offers theoretical energy density five times higher than conventional lithium battery technologies. While challenges with cycle life and sulfur utilization remain, advances in cathode design and electrolyte formulation are bringing this technology closer to commercialization.
Redox Flow Batteries
For large-scale energy storage, lithium-based redox flow batteries separate energy storage (in liquid electrolytes) from power generation (in a stack), offering scalability and long cycle life advantages for grid-scale lithium battery applications.

Research laboratory developing next-generation lithium battery technologies
These innovations aim to address the key challenges facing current lithium battery technology, including energy density limitations, charging speed, safety concerns, and raw material constraints. As research progresses, we can expect lithium battery performance to continue improving while costs decline, enabling broader adoption in electric transportation, renewable energy integration, and portable electronics.Optical Transceiver.
Conclusion
The lithium battery represents a remarkable convergence of electrochemistry, material science, and engineering design. Its ability to reversibly store and deliver electrical energy through controlled lithium ion migration has transformed modern technology.Electronic shelf labels.
As demand for energy storage continues to grow—driven by electric vehicles, renewable energy integration, and portable electronics—understanding the fundamental principles of the lithium battery becomes increasingly important. Ongoing research promises to further enhance lithium battery performance, making it an even more critical component of our sustainable energy future.Related Hydraulic Spare Parts.